In this lesson, students apply what they’ve learned about the variations in the water cycle to explore how evaporation affects ocean water density, which in turn affects weather and climate.
In this unit, students investigate the relationship between the water cycle, ocean salinity, weather, and climate. In this lesson, students explore ocean salinity and analyze how ocean density is related to its molecular structure. This page highlights key components of this lesson.
Science background gives teachers more in-depth information on the phenomena students explore in this unit. Here is an excerpt from this section on ocean salinity and density.
All water eventually ends up in the ocean, where it deposits the salt and other matter. Over millions of years, this salt has built up, creating the salt water of the oceans. But oceans do not all have the same concentration of salt. The data gathered by the Aquarius satellite reveal patterns in ocean salinity: salinity is low in areas where precipitation is greater than evaporation, mainly in regions near the coast and near the equator. In contrast, salinity is often high in the middle of the ocean, away from rivers, which release fresh water into the oceans.
The cause of these patterns in salinity can be understood by first thinking about the basic structure of salt water. Salt water is a mixture, which means it is made up of two or more pure substances that are mixed together but not chemically bonded. Salt water is made up of different kinds of chemicals that make it salty. The most common is salt (NaCl), which is a molecule made up of one atom of sodium and one atom of chlorine. Water (H2O) is a molecule made up of two atoms of hydrogen and one atom of oxygen.
Because salt water is a mixture, the parts that make it up can be separated. Imagine you leave a cup of salt water on a table next to a sunny window. Over time, the water will evaporate, leaving behind the salt. This is because salt cannot evaporate in the same way that water does.
The variations of the water cycle around the planet play a significant role in the differing concentrations of salt in the world’s oceans. When ocean water freezes into ice or evaporates, the remaining ocean water becomes saltier because there is less liquid water relative to the amount of salt present.
This causes the salt water to become denser. Remember that density is the measure of the amount of mass in a given volume. The more tightly packed the molecules of a substance are, the denser the substance is because it has more mass in a given space than do materials that are less dense. The more salt there is packed into the water, the denser the water is. In contrast, precipitation decreases the salinity of ocean water because precipitation is always fresh water, so when it falls into the ocean, it dilutes the concentration of salt. Because of this, parts of the ocean that receive a lot of rain often have low salinity. Similarly, rivers are fresh water, so areas of the ocean that are near mouths of rivers that empty fresh water into the ocean are less salty. Both of these inputs of fresh water decrease salinity and make the water less dense.
The temperature of water also affects its density. As water gets warmer, its molecules spread out, so it becomes less dense. As it gets colder, it becomes denser because its molecules clump together. (It is important to note that unlike most substances, solid ice is actually less dense than liquid water. This has to do with the unique properties of the water molecule and how the atoms bond together. This is why ice floats on water.)
In this lesson, students apply what they’ve learned about the variations in the water cycle to explore how evaporation affects ocean water density, which in turn affects weather and climate.

Prepared hands-on materials, full year grade-specific curriculum, and personalized live professional development designed to support mastery of current state science standards.
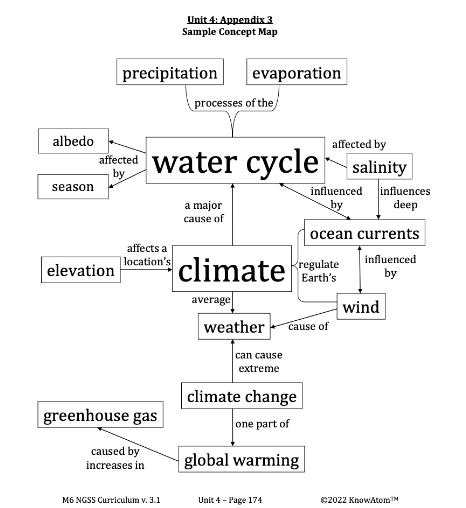
Climate : the average weather in a location over 30 years or more
Evaporation : the process of liquid water changing into water vapor, its gas state
Ocean Currents : paths of flowing ocean water that push warm and cold water to different parts of the planet
Precipitation : the process of water falling back to Earth in the form of rain, snow, sleet, or hail
Salinity : the measure of all the salts dissolved in water
Season : a period of time characterized by specific weather patterns and by the length of day and night
Water Cycle : the circulation of water through the hydrosphere from Earth’s surface to the atmosphere and back
Weather : the conditions of the atmosphere (temperature, humidity, wind speed, and precipitation) at a particular place and time

Studying the Ocean’s Saltiness
Since 2011, a satellite called Aquarius has been orbiting Earth from 644 kilometers (400 miles) above the surface. Its mission has been to gather data about the salinity in the surface seawater. Salinity is the measure of all the salts dissolved in water. If you’ve ever gone swimming in the ocean and accidently swallowed some water, you’ve tasted how salty the ocean is.
For hundreds of years, scientists physically measured the salinity of different parts of the ocean from ships and buoys. This provided limited data, however. Some parts of the ocean were much harder for scientists to reach and measure than others. In response to this problem, a team of about 100 scientists and engineers developed and launched the Aquarius satellite in 2011. The Aquarius satellite improves on older methods of gathering data because it continuously monitors and collects data on ocean salinity around the planet.
Understanding the ocean’s salinity, including how that salinity differs around the world and changes over time, is essential for scientists who want to understand the movement of water around the planet. This in turn has major implications for weather and climate. Weather is the conditions of the atmosphere (temperature, humidity, wind speed, and precipitation) at a particular place and time. Climate is the average weather in a location over 30 years or more.
How Oceans Get Salty
Understanding salinity first begins with an understanding of how oceans become salty. Oceans become salty because of interactions among different Earth systems. Remember that a system is a set of connected, interacting parts that form a more complex whole. Systems have inputs and outputs. Inputs are what are received by the system. Outputs are what are sent from the system.
Earth has four primary systems that are interconnected, constantly interacting with and influencing one another—the hydrosphere, atmosphere, biosphere, and geosphere.
Earth’s systems are interconnected, constantly interacting with and influencing one another. For example, all of the water on Earth forms an Earth system called the hydrosphere. The hydrosphere includes all of the ice, liquid water, and water vapor on Earth. It is constantly interacting with another Earth system called the atmosphere. The atmosphere is the mixture of gasses, dust, water vapor, and other molecules above Earth’s crust.
One of the gasses in the atmosphere is carbon dioxide. When water falls to Earth’s surface as rain, it carries some of this carbon dioxide, making the water slightly acidic. This slightly acidic water breaks down the rocks on Earth’s surface into smaller particles, including salt and other minerals. This is an interaction between the hydrosphere and the geosphere—the Earth system made up of Earth’s landforms, including mountains, valleys, soil, and sediment.
All water that collects on Earth’s surface will eventually flow downhill to the oceans because of the pull of Earth’s gravity. As water flows downhill, it carries with it the particles of salt and other matter. All water eventually ends up in the ocean, where it deposits the salt and other matter. Over millions of years, this salt has built up in the oceans, creating the salt water of the oceans.


Why Salinity Matters
Ray Schmitt is the lead scientist on a scientific mission that is using data from the Aquarius satellite. Schmitt has long been fascinated by salt in the ocean. He was one of the first scientists to recognize how important the ocean is in the water cycle.
In 2012, Schmitt and a team of scientists set sail for the world’s saltiest patch of open ocean water, located halfway between the Bahamas and the western coast of North Africa. Scientists say this spot is similar to a desert on land, where there is little rainfall and a lot of evaporation. Data suggest that this spot of ocean is actually becoming saltier over time.
The scientists on board the ship stayed for three weeks, taking measurements on the ocean’s salinity, temperature, and other factors. They used many different kinds of scientific tools, including floats, gliders, drifters, moorings, ships, satellites, and computer models. One of the reasons that scientists care about variations in ocean salinity is that they play a major role in deep ocean currents. Ocean currents are paths of flowing ocean water that push warm and cold water to different parts of the planet.
Cold, dense water in the oceans sinks deep and spreads out all around the world. The sinking water is replaced by the warm, less-dense water near the surface that moves to the north. Scientists call this the Great Ocean Conveyor Belt.
For the hands-on activity, students carry out an experiment to explore the effect of evaporative water loss on ocean density. Students calculate the density of salt water in an open system and a closed system. They then expose the open condition to evaporative wind over a few days, and compare the change in density that occurs in both systems. Students use the data from the experiment to analyze the relationship between the amount of water evaporation and the density of the water.
KnowAtom incorporates formative and summative assessments designed to make students thinking visible for deeper student-centered learning.
Standards citation: NGSS Lead States. 2013. Next Generation Science Standards: For States, By States. Washington, DC: The National Academies Press. Neither WestEd nor the lead states and partners that developed the Next Generation Science Standards were involved in the production of this product, and do not endorse it.
